Crispy, brown toast and grill marks on your chicken, fish or beef both contain elevated amounts of Advanced Glycation End (AGE) Products, a group of molecules formed during high temperature cooking (greater than 250ºF; i.e. frying, grilling, roasting, baking). In a previous article (https://michaellustgarten.wordpress.com/2014/07/25/advanced-glycation-end-products-theres-more-to-health-than-counting-calories-protein-fat-and-carbs/) I wrote about the adverse health effects of consuming a high AGE product diet (measured as carboxymethyl-lysine, CML), including increased body weight (without increasing food intake), increased oxidative stress and oxidative damage, insulin resistance, and, a reduced lifespan (Cai et al. 2007). In a second study, the lifespan extending effects of calorie restriction (the gold standard in terms of extending lifespan) were abolished by adding the AGE products CML and methylglyoxal (MG) back into the diet (Cai et al. 2008). However, these results are in rodents- what does the evidence say in people?
Unfortunately, in people the story does not get better. AGE products have been shown to play a causative role in atherosclerosis, diabetes, cardiovascular disease, and, chronic kidney disease (Semba et al. 2010). And, it gets worse-older adults with elevated levels of CML are at greater risk for arterial stiffness (Semba et al. 2009), anemia (Semba et al. 2008), poor muscle strength (Dalal et al. 2009), low physical performance (Semba et al. 2010), and, increased risk of all-cause mortality (Semba et al. 2009). Not good!
So, the take home message is to not eat food browned at a high temperature, and, to reduce the amount of AGE products consumed in the diet, right? After all, the contribution of dietary AGE products to the total pool of AGE products in the body is much greater than the amount of AGE products that are endogenously generated by abnormal glucose metabolism or lipid oxidation (Henle 2003).
But, vegans and vegetarians have been shown to have higher levels of the AGE product CML in their blood, relative to meat-eaters (Sebeková et al. 2001, Krajcovicová-Kudlácková et al. 2002)! And, obese children have lower levels of CML than lean children (Sebeková et al. 2009). Furthermore, people with high levels of fat have been shown to have lower levels of CML, and vice versa-those with low body fat have been shown to have high levels of CML (Semba et al. 2011)! In other words, populations that are well documented to have improved health, including vegans, vegetarians and lean subjects have elevated blood levels of the same AGE product, CML that has been shown to be either associated with or that cause all sorts of bad things in both rodents and people. Does that mean that vegans, vegetarians and lean subjects will eventually suffer from the same AGE product induced ill-health? Let’s dissect the data…
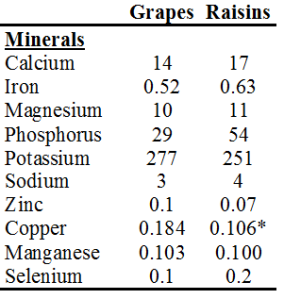
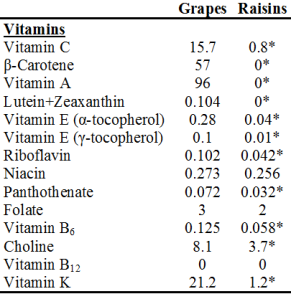
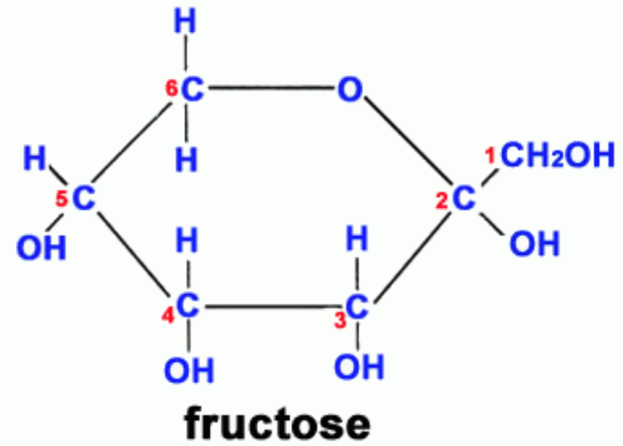
Study #1: AGE products (measured as CML and fluorescence) were shown to be higher in vegans, lacto-ovo-vegetarians and semi-vegetarians, relative to omnivores. Study strengths: young subjects, between 30-40y (thereby minimizing the confounding affect of age on AGE product formation), and, all groups were within a healthy BMI range (<25 kg/m2). As expected, the BMI for vegans and vegetarians was significantly lower than found in omnivores. All subjects had been on these diets for 7-8 years. Weakness: small study population-9 vegans, 19 lacto-ovo-vegetarians, 14 semi-vegetarians, 19 omnivores. The authors hypothesized that higher fruit intake by vegans and vegetarians might be the reason for the elevated CML found in these groups (Sebekova et al. 2001). CML is primarily formed via the reaction of fructose (the main sugar found in fruit) with the amino acid lysine (Figure 2). In support of this, AGE products (measured by fluorescence) have been shown to form at a greater rate in the presence of fructose, relative to glucose (Jakus et al. 1998).
Study #2: Plasma CML and fluorescent AGE products were found to be higher in vegetarians, relative to meat eaters. As in study#1, a lower BMI was reported for vegetarians (22 kg/m2), relative to meat-eaters (24 kg/m2), indicating that this was a healthy cohort. Also, young subjects in both groups-on average, 31y for meat-eaters and 36y for vegetarians. However, a limited number of total study subjects, 38, were used. In support of the hypothesis from study#1 that a higher fruit intake was responsible for the elevation in CML found in vegetarians, the frutose/lysine ratio was found to be higher in vegetarians, compared with meat-eaters (Krajcovicová-Kudlácková et al. 2002).
Study #3: Overweight children (BMI 27 kg/m2, compared with 22 kg/m2 for controls; average age, 12y) had significantly reduced (~30% ) concentrations of plasma AGE product fluorescence, fructose-lysine, and CML than their lean counterparts, despite being insulin resistant, having elevated inflammation, and, elevated protein and DNA oxidation (Sebeková et al. 2009).
Study #4: Serum CML was found to be inversely associated with total fat mass, trunk fat mass, and fat mass in the arms + legs combined, after adjusting for age, sex, BMI, systolic blood pressure, TG, HDL cholesterol, and renal function (Semba et al. 2011). In other words, those with high CML were found to have have low fat mass, whereas those with low CML have high fat mass.
Study #5: No difference in serum CMLwas found when comparing obese with lean subjects; CML was reported to be elevated in fat tissue from obese subjects (Gaens et al 2007).
What’s going on here? Leanness is well documented as associated with improved health, yet, in these studies, lean subjects had higher CML relative to those with higher fat mass. In addition, two separate studies showed higher CML in healthy vegans and vegetarians, compared with meat eaters! How is this possible?
First, use of fluorescence to detect products related to oxidative stress has been problematic (Muller 2009), as auto-oxidation of the probes commonly used for detection often makes the measurement unreliable. But what about CML? There is a wealth of data detailing the adverse effects of CML on both rodent and human health. In my opinion, this is a perfect example of correlation not proving causation. In most of the studies that measure AGE products, CML is used as the global marker of AGE product content. Because the data in healthy populations (vegans, vegetarians, lean subjects) contradicts the wealth of CML related evidence in terms of health, something must be wrong in using CML as a marker of AGE products, and, overall health. In other words, CML is associated with many adverse outcomes, but does not prove it. In support of this, in one of the rodent studies that showed a reduction in lifespan that was associated with increased CML, an additional AGE product, methylglyoxal was also associated with this adverse outcome. Methylglyoxal has been shown to increase inflammation (TNF-α production) and oxidative stress (glutathione depletion) to a much greater degree than CML (Cai et al. 2002). Although both methylglyoxal and CML have both been shown to increase during aging (Uribarri et al 2007), methylglyoxal is not commonly measured in relation to the adverse outcomes found in humans.
So, if you’re lean, vegan or vegetarian and you have high levels of CML, should you worry that you’re on the road to ill-health? The easy answer is no, but, in my opinion, if your methylglyoxal levels are also high, it might be time to stop/minimize eating foods that have been cooked at a high temperature…
If you’re interested, please have a look at my book!
References:
Cai W, Gao QD, Zhu L, Peppa M, He C, Vlassara H. Oxidative stress-inducing carbonyl compounds from common foods: novel mediators of cellular dysfunction. Mol Med. 2002 Jul;8(7):337-46.
Cai W, He JC, Zhu L, Chen X, Wallenstein S, Striker GE, Vlassara H. Reduced oxidant stress and extended lifespan in mice exposed to a low glycotoxin diet: association with increased AGER1 expression.Am J Pathol. 2007 Jun;170(6):1893-902.
Cai W, He JC, Zhu L, Chen X, Zheng F, Striker GE, Vlassara H. Oral glycotoxins determine the effects of calorie restriction on oxidant stress, age-related diseases, and lifespan. Am J Pathol. 2008 Aug;173(2):327-36.
Dalal M, Ferrucci L, Sun K, Beck J, Fried LP, Semba RD. Elevated serum advanced glycation end products and poor grip strength in older community-dwelling women. J Gerontol A Biol Sci Med Sci. 2009;64:132-137.
Gaens KHJ, van de Waarenburg MPH, Nijhuis J, Scheijen J, Stehouwerr CDA, Schalkwijk CG. Increased formation of N-(carboxymethyl)lysine (CML) in human adipose tissue; possible biological consequences. 9th International Symposium on the Maillard Reaction, 2007. Abstract.
Henle T. AGEs in foods: do they play a role in uremia? Kidney Int. 2003; Suppl 84:S145-7.
Jakus V, Rietbrock N, Hrnciarová M (1998) Study of inhibition of protein glycation by fluorescence spectroscopy. Chem Papers 52:446
Krajcovicová-Kudlácková M, Sebeková K, Schinzel R, Klvanová J. Advanced glycation end products and nutrition. Physiol Res. 2002;51(3):313-6.
Muller FL. A critical evaluation of cpYFP as a probe for superoxide. Free Radic Biol Med. 2009 Dec 15;47(12):1779-80.
Sebeková K, Somoza V, Jarcusková M, Heidland A, Podracká L. Plasma advanced glycation end products are decreased in obese children compared with lean controls. Int J Pediatr Obes. 2009;4(2):112-8.
Sebeková K, Krajcoviová-Kudlácková M, Schinzel R, Faist V, Klvanová J, Heidland A. Plasma levels of advanced glycation end products in healthy, long-term vegetarians and subjects on a western mixed diet. Eur J Nutr. 2001 Dec;40(6):275-81.
Semba RD, Patel KV, Sun K, Guralnik JM, Ershler WB, Longo DL, Ferrucci L. Association of serum carboxymethyl-lysine, a dominant advanced glycation end product, with anemia in adults: the Baltimore Longitudinal Study of Aging. <i>J Am Geriatr Soc</i>. 2008;56:2145-2147.
Semba RD, Bandinelli S, Sun K, Guralnik JM, Ferrucci L. Plasma carboxymethyl-lysine, and advanced glycation end product, and all-cause and cardiovascular disease mortality in older community-dwelling adults. <i>J Am Geriatr Soc</i>. 2009;57:1874-1880.
Semba RD, Najjar SS, Sun K, Lakatta EG, Ferrucci L. Serum carboxymethyl-lysine, an advanced glycation end product, is associated with increased aortic pulse wave velocity in adults. Am J Hypertens. 2009;22:74-79.
Semba RD, Nicklett EJ, Ferrucci L. Does accumulation of advanced glycation end products contribute to the aging phenotype? J Gerontol A Biol Sci Med Sci. 2010;65:963-75.
Semba RD, Bandinelli S, Sun K, Guralnik JM, Ferrucci L. Relationship of an advanced glycation end product, plasma carboxymethyl-lysine, with slow walking speed in older adults: the InCHIANTI study. <i>Eur J Appl Physiol</i>. 2010;108:191-195.
Semba RD, Fink JC, Sun K, Windham BG, Ferrucci L. Elevated serum advanced glycation end products are associated with renal insufficiency: the Baltimore Longitudinal Study of Aging. J Ren Nutr. 2010;20:74-81.
Semba RD, Arab L, Sun K, Nicklett EJ, Ferrucci L. Fat mass is inversely associated with serum carboxymethyl-lysine, an advanced glycation end product, in adults. J Nutr. 2011 Sep;141(9):1726-30.
Uribarri J, Cai W, Peppa M, Goodman S, Ferrucci L, Striker G, Vlassara H. Circulating glycotoxins and dietary advanced glycation endproducts: two links to inflammatory response, oxidative stress, and aging. J Gerontol A Biol Sci Med Sci. 2007;62:427-33.