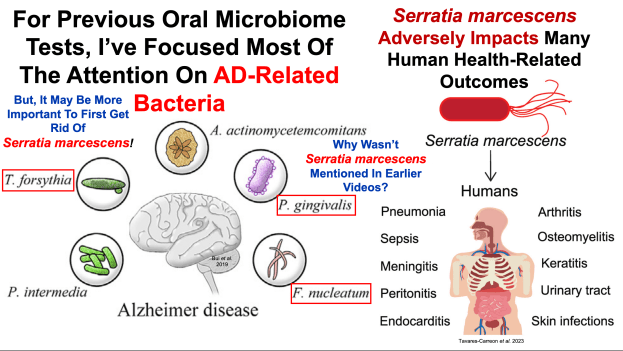
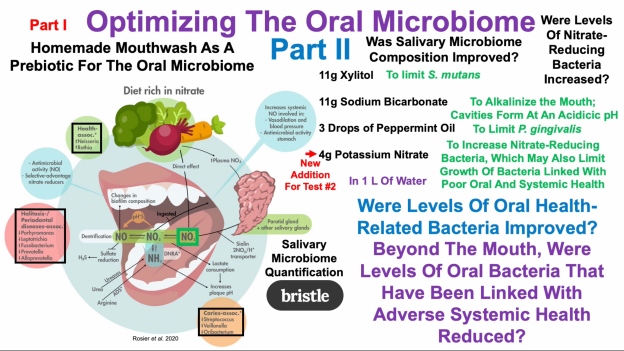
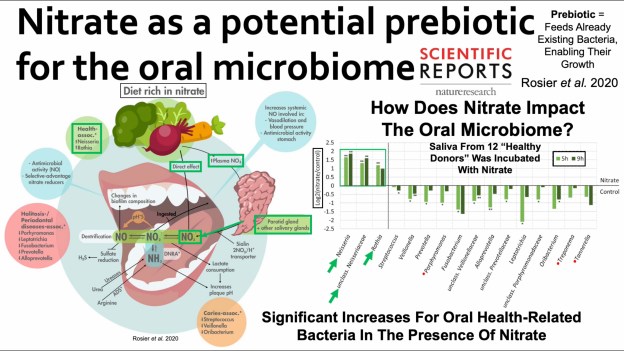
I’d like to keep my teeth healthy for as long as possible. I brush my teeth in the morning and at night, and floss every night, because daily flossing is associated with reduced mortality risk (https://atomic-temporary-71218033.wpcomstaging.com/2014/07/23/dental-floss-cancer-and-mortality-is-there-an-association/). Is there anything else that I can do to optimize my oral health? To answer that question, it’s important to know how tooth decay (also known as caries) occurs. As shown below, bacteria located on our teeth (in plaque) use dietary sugars to make acid, which demineralizes tooth enamel, thereby resulting in caries (Limeback et al. 2012).

Not all dietary sugars are equal in their ability to produce tooth decay. In the picture below, sugars with red boxes are the most cariogenic. For example, the worst offenders in terms of tooth decay are sucrose and glucose. Also note that starch is cariogenic, but less when compared with sucrose and glucose. The least cariogenic sugars are sorbitol and mannitol, and, the sugar xylitol has been shown to be protective against tooth decay (Limeback et al. 2012).

After eating a high sugar meal, there is an oral bacteria-induced increase in acidity that can lead to tooth demineralization. At an oral plaque pH of 5.5, tooth enamel demineralization occurs (Stephan and Miller 1943). Shown below is a graph of plaque pH vs. time for the 60 minutes following an oral glucose rinse. The green line is indicative of someone who is caries resistant: their plaque pH decreases from ~6.8 to ~5.8, followed by a relatively quick return to the starting pH value after 60 minutes. Note that at no point does the green line drop below the 5.5 threshold for enamel demineralization to occurr. The yellow line is indicative of someone at moderate tooth decay risk: their plaque pH drops from 6.8 to below 5.5, where it remains for ~ 10 minutes, and then slowly returns to baseline pH values after 60 minutes. The red line is indcative of someone that is at high risk for tooth decay: after the glucose rinse at time 0, their plaque pH rapidly drops below 5.5 and remains there for ~50 minutes, where significant teeth demineralization may occur. Note that their plaque pH doesn’t return to the baseline pH value after 60 minutes, either.

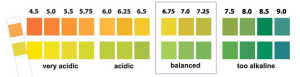
With these data in mind and my goal to reduce tooth decay risk I bought pH strips to test my daily oral pH. Shown below is what the pH strip looks like, without having been dipped in my saliva. Adjacent to that is the pH color scale, and to the right, my average daily fasted oral pH, ~6.5:


The good news is that I’ve yet to see an oral pH value below 6.5, which puts me at low risk for tooth decay. To further minimize that risk, in addition to brushing and flossing, after meals and throughout the day I gargle with a homemade 1% sodium bicarbonate (physiological saline is ~0.9%) and 10% xylitol solution. I use sodium bicarbonate to temporarily neutralize any potential oral acids. Xylitol can protect against tooth decay by reducing plaque and the level of tooth decay-causing bacteria (i.e. Streptococci mutans) in saliva and plaque (Söderling 2009). For example, fluoride-containing toothpaste in the presence of 10% xylitol reduces tooth decay more than fluoride toothpaste alone (Sintes et al. 1995; Sintes et al. 2002). However, it’s important to note that some xylitol-based studies have not been shown to reduce tooth decay risk (Riley et al. 2015). At worst, including xylitol may have no effect, whereas at best it may reduce oral Streptococci mutans, thereby decreasing tooth decay risk.
After gargling, my normal oral ph (~6.5) changes to alkaline (~8.5), as shown in the pH strip below:

Couldn’t I use a store-bought mouthwash instead? The measured pH of several commercially available mouthwashes is shown in the table below (Sun et al. 2014). Note that some are below the critical pH for tooth demineralization, 5.5! Others aren’t too far away from 5.5, either. In contrast, the pH of my homemade mouthwash pH is ~8.0.

If you’re interested, please have a look at my book!
References
Limeback H et al. Comprehensive Preventive Dentistry, Chapter 1: “A brief introduction to oral diseases: caries, periodontal disease, and oral cancer”. page 1-24. July 2012. ISBN: 978-0-8138-2168-9.
Riley P, Moore D, Ahmed F, Sharif MO, Worthington HV. Xylitol-containing products for preventing dental caries in children and adults. Cochrane Database Syst Rev. 2015 Mar 26;3:CD010743.
Sintes JL, Escalante C, Stewart B, McCool JJ, Garcia L, Volpe AR, Triol C. (1995). Enhanced anticaries efficacy of a 0.243% sodium fluoride/10% xylitol/silica dentifrice: 3-year clinical results. American Journal of Dentistry, 8, 231–235.
Sintes JL, Elías-Boneta A, Stewart B, Volpe AR, Lovett J. (2002). Anticaries efficacy of a sodium monofluorophosphate dentifrice containing xylitol in a dicalcium phosphate dihydrate base. A 30-month caries clinical study in Costa Rica. American Journal of Dentistry, 15, 215–219.
Söderling EM. (2009) Xylitol, mutans streptococci, and dental plaque. Advances in Dental Research, 21, 74–78.
Stephan RM and Miller BF. (1943) A Quantitative Method for Evaluating Physical and Chemical Agents which Modify Production of Acid in Bacterial Plaques on Human Teeth. Journal of Dental Research, 22, 45–51.
Sun FC, Engelman EE, McGuire JA, Kosmoski G, Carratello L, Ricci-Nittel D, Zhang JZ, Schemehorn BR, Gambogi RJ. Impact of an anticaries mouthrinse on in vitro remineralization and microbial control. Int J Dent. 2014;2014:982071.