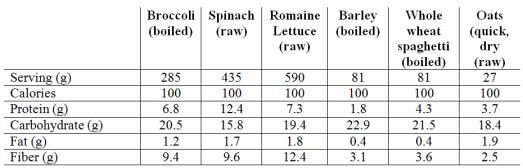
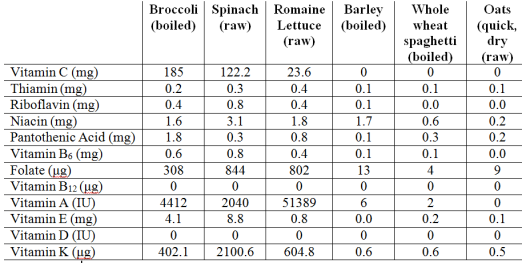
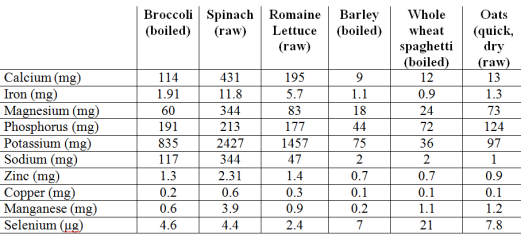
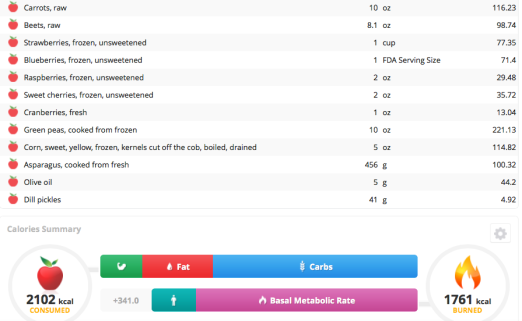
My approach to optimizing health and potentially lifespan includes daily nutrient tracking and yearly blood testing. Once you get your blood test results back from the doctor, are your values optimal if you’re within the reference range? This article will examine the “optimal range” for 2 of these measurements, serum bicarbonate and the anion gap.
What does serum bicarbonate measure? The amount of bicarbonate in the blood is indicative of dietary acid load (Adeva and Souto 2011), systemic metabolism, lung and kidney function. First, a diet rich in animal products and grains (acid-forming), and poor in fruits and vegetables (base-forming) can induce a state of metabolic acidosis (Sebastian et al. 2001). Similarly, cellular metabolism produces carbon dioxide (CO2), a gas that is an acid. The lungs and kidneys act to remove systemic increases in acid load: CO2 reacts with water to form bicarbonate (H2CO3-), where it travels to the lung for excretion by exhaling it as CO2. The kidneys decrease acid (whether from the diet or metabolism) by removing protons (H+) from the blood, followed by urinating the acid out of the body, and also by producing bicarbonate. In sum, serum bicarbonate is a measure of acid load: from the diet, by your body’s ability to produce it, by your kidney’s ability to buffer it, and by your lungs ability to remove it.
Low serum bicarbonate is indicative of increased systemic acidity, whereas a high serum bicarbonate indicates systemic alkalinity. If systemic acidity is high, bicarbonate will be consumed to neutralize the acid, thereby decreasing serum bicarbonate. Assuming that bicarbonate is not being consumed in the diet (via fruits and vegetables), the kidney would have to then produce bicarbonate to make up for the increase in bicarbonate consumption.
The anion gap is a second indicator of systemic acid/base balance. It is a measure of the positively and negatively charged ions in blood, and includes serum bicarbonate. It is calculated by adding the serum concentrations of sodium (Na) and potassium (K), while subtracting chloride (Cl-) and bicarbonate (HCO3-):
Anion gap = ( [Na+] + [K+] ) − ( [Cl−] + [HCO3−] )
A high anion gap is indicative of systemic acidity whereas a low value is indicative of alkalinity.
The reference range for serum bicarbonate and the anion gap are 20-30 and 5-18 mEq/L. On my latest blood test (8/2015) my values were 31 and 6, respectively…Are these values optimal for health?
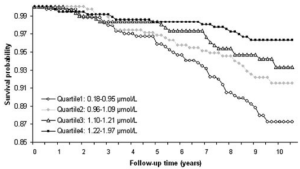
First, as shown below, decreased serum bicarbonate values are associated with increased risk for future physical function limitation (Yenchek et al. 2014). In a study of 1544 overweight (BMI ~27 kg/m2) older adults (average age, ~75 years) with a median follow-up of ~4 years, acidic serum bicarbonate values (less than 25.9) had an increased risk for future functional limitation, when compared with subjects with more alkaline values (greater than 26). It is important to note that age-related decreased kidney function leads to an inability to produce bicarbonate, thereby decreasing serum bicarbonate. However, after adjusting for the presence or absence of subjects with chronic kidney disease (CKD), the association between a more acidic serum bicarbonate value with future functional limitation remained. In other words, poor kidney function was not driving the effect of acidosis on risk for future functional limitation.
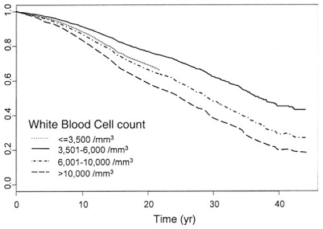
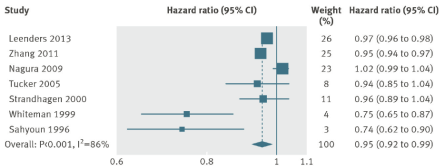
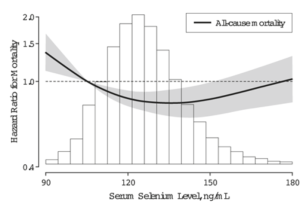
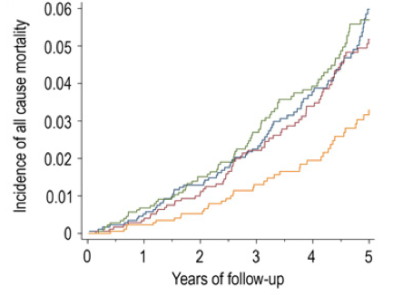
In a larger study that included 31,590 subjects with average age of ~50 years, an average BMI <25 kg/m2, and a median follow up ~8 years, a serum bicarbonate value < 26, compared with 31, had a 46% significantly increased all-cause mortality risk (see below; Park et al. 2015). For the anion gap, although mortality risk was increased at values > 11, compared with less than 6, this finding was not statistically significant. Nonetheless a trend for increased mortality risk with a more acidic value for the anion gap was present. In addition, although urine pH is not generally measured when you get a yearly physical, it’s an easy (just pee in a cup!) and inexpensive way to see if you’re peeing out more acid or base. In the figure below, we see that with urinating out more base (pH >8.0) as the reference, peeing out more acid (pH <7.5) is associated with a ~250% increased mortality risk! Assuming you have properly functioning kidneys, urinating more base will happen if your diet is rich in alkaline-rich foods, like vegetables. In contrast, a high meat and grains-based diet will lead to urinating out more acid.
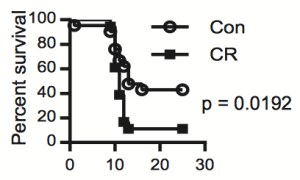
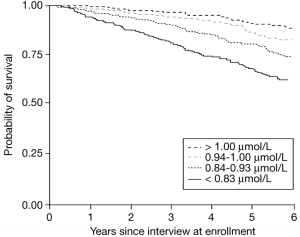
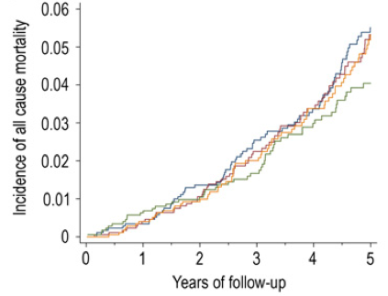
In contrast to these data, shown below are the findings of Raphael et al. 2013, who found no association between serum bicarbonate with mortality risk. In that study, 15,836 overweight (the BMI range average was from 26-29) subjects with an average age ~43 years were followed for ~9 years. Although an acidic serum bicarbonate value (<22, compared with 26-30 as the reference) was associated with a 75% increased all-cause mortality risk, when excluding subjects with CKD from the analysis, that association was no longer statistically significant. However, it is important to note a similar trend (albeit non-significant) of association between acidic serum bicarbonate values with an increased mortality risk was present in those that did not have CKD.
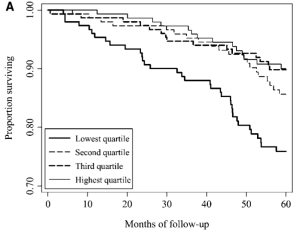
Further support for alkaline values for serum bicarbonate or the anion gap being beneficial for health is shown below. A low anion gap (after adjusting for serum albumin) was associated with better survival with a more alkaline value (<10.5), when compared with acidic values (>10.5) in 862 normal weight (BMI ~24) elderly (average age ~74y), during a 5-year follow up (Ahn et al. 2014). Included in the improved survival rate were decreases in cardiovascular disease and infection-related mortality.
One criticism of this data is that these associations are in older adults, and that age-related decreases in kidney function may lead to an inability to produce bicarbonate, thereby increasing the anion gap. In disagreement with that critique, young subjects (age range, 20-49 years) with low serum bicarbonate and an elevated anion gap (which together suggest systemic acidosis) were significantly more likely to have decreased cardiorespiratory fitness (VO2 max) (Abramowitz et al. 2012).
Collectively, based on these data it looks like my serum bicarbonate (31) and anion gap (6) values are close to optimal for health and longevity. If your values are not close to optimal, can they be improved? Yes! Increasing fruit and vegetable (F&V) intake has been shown to increase serum bicarbonate (Goraya et al. 2013). Because bicarbonate is a component of calculating the anion gap, an increase in fruit and vegetable intake would be expected to also decrease the anion gap (although I couldn’t find any studies that have tried to use F&V to reduce it).
If you’re interested, please have a look at my book!
References
Abramowitz MK, Hostetter TH, Melamed ML. Lower serum bicarbonate and a higher anion gap are associated with lower cardiorespiratory fitness in young adults. Kidney Int. 2012 May;81(10):1033-42.
Adeva MM, Souto G. Diet-induced metabolic acidosis. Clin Nutr. 2011 Aug;30(4):416-21.
Ahn SY, Ryu J, Baek SH, Han JW, Lee JH, Ahn S, Kim KI, Chin HJ, Na KY, Chae DW, Kim KW, Kim S. Serum anion gap is predictive of mortality in an elderly population. Exp Gerontol. 2014 Feb;50:122-7.
Goraya N, Simoni J, Jo CH, Wesson DE. A comparison of treating metabolic acidosis in CKD stage 4 hypertensive kidney disease with fruits and vegetables or sodium bicarbonate. Clin J Am Soc Nephrol. 2013 Mar;8(3):371-81.
Park M, Jung SJ, Yoon S, Yun JM, Yoon HJ. Association between the markers of metabolic acid load and higher all-cause and cardiovascular mortality in a general population with preserved renal function. Hypertens Res. 2015 Jun;38(6):433-8.
Raphael KL, Zhang Y, Wei G, Greene T, Cheung AK, Beddhu S. Serum bicarbonate and mortality in adults in NHANES III. Nephrol Dial Transplant. 2013 May;28(5):1207-13.
Sebastian A, Frassetto LA, Sellmeyer DE, Merriam RL, Morris RC Jr. Estimation of the net acid load of the diet of ancestral preagricultural Homo sapiens and their hominid ancestors. Am J Clin Nutr. 2002 Dec;76(6):1308-16.