Our story begins with Michael Rose, who used an experimental evolution approach to breed flies that live more than 2-fold longer than controls (Rose 1999). In contrast with the one gene at a time knockout or overexpression strategy that is ubiquitous in modern biology, Rose separated initially genetically homogeneous flies into two groups-one with delayed and the other with normal reproduction. As shown below, after 80 generations, the group with continually delayed reproduction had ~2-fold increased average and maximal lifespan.
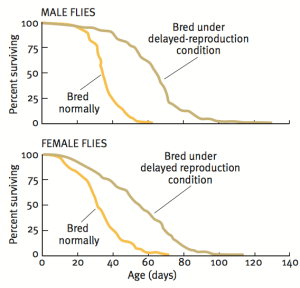
While this suggests that continually delaying reproduction may extend lifespan in people, the time it would take to do that makes it an unreasonable strategy. In 2010, the average age at first reproduction in the US was 25.4 years (http://www.cdc.gov/nchs/data/nvsr/nvsr61/nvsr61_01.pdf). Therefore, to replicate the doubling of lifespan found in Drosophila, 80 generations * 25.4y would take more than 2000 years! In contrast, a more reasonable approach towards extending life in people may involve stimulation of some of the pathways involved in the extended fly lifespan.
What genetic mechanisms underlie this 2-fold increase in Drosophila lifespan? Kurapati et al. 2000) found that levels of the mitochondrially located heat shock protein 22 (Hsp22) were between two and ten-fold higher in long-lived Drospohila, relative to the shorter-lived controls. In 2004 Morrow et al. identified the causative role of Hsp22 overexpression, as average lifespan increased by ~30%, thereby implicating mechanisms related to upregulation of Hsp22 on increasing lifespan.
Unfortunately, the role of Hsp22 on influencing lifespan in mammals is unknown. But, can we learn something about the underlying mechanism of the Hsp22-induced increase in lifespan and apply that to mammalian aging?
Hsp22 expression is regulated by histone deacetylase (HDAC) inhibitors (Zhou et al. 2005). In other words, when certain HDAC’s are inhibited, histone acetylation increases, resulting in elevated Hsp22 expression. Interestingly, histone acetylation has also been shown to be involved in lifespan determination in yeast and worms (Kaeberlein et al. 1999, Kang et al. 2002, Kim et al. 1999, Tissenbaum and Guarente, 2001). In contrast, the HDAC’s that have been popularized by the resveratrol-sirtuin story results in histone deacetylation, or the removal of acetyl groups from histones.
Identification of compounds that inhibit the HDAC’s that control Hsp22 expression would seem to be a good method for potentially increasing mammalian lifespan. Supplementation with sodium butyrate increases Hsp22 expression in Drosophila (Zhao et al. 2005), resulting in increased Drosophila lifespan (McDonald et al. 2013). Interestingly, sodium butyrate is a class I, II, IV HDAC inhibitor, whereas the sirtuins are class III inhibitors (Witt et al. 2009), evidence that suggests differing roles for the HDACs on lifespan extension.
Unfortunately, the causative role of butyrate-producing bacteria on mammalian lifespan has yet to be directly tested. However, butyrate stimulates expression of fibroblast growth factor 21 (Li et al. 2012), a protein whose overexpression extends both average and maximal lifespan in mice (shown below; Zhang et al. 2012).

Furthermore, acarbose supplementation extends median and maximal lifespan in genetically heterogeneous mice (Harrison et al. 2013), an important finding because acarbose supplementation has been shown to elevate serum butyrate in human subjects (Wolever and Chiasson 2000).
How can we get butyrate into our diet? Although butter contains small amounts of butyrate, a butter-rich diet has been shown to be obesogenic (Hariri et al. 2010). Fortunately, there is another way we can increase levels of butyrate, and that’s by stimulating our intestinal bacteria to produce it! The most abundant butyrate-producing gut bacterial species are Faecalibacterium prausnitzii, Eubacterium rectale, Eubacterium hallii and Anaerostipes hadrus (Tap et al. 2009, Walker et al. 2011).
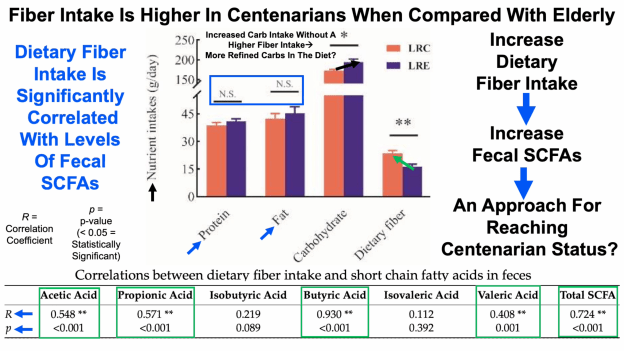
Interestingly, butyrate-producing bacteria decrease during aging, which, in my opinion makes colonizing our intestines with these beneficial bacteria all the more important. For example, Faecalibacterium prausnitzii, Eubacterium hallii and Eubacterium rectale are significantly reduced in centenarians when compared with elderly and young subjects (Biagi et al. 2010). Several other gut bacterial butyrate producers are also reduced in centenarians when compared with the other age groups, including Ruminococcus obeum, Roseburia intestinalis, E. ventriosum, and, Papillibacter cinnamovorans.
Collectively, these data suggest that increasing gut bacterial species that produce butyrate may be important for increasing lifespan in both lower organisms and, in mammals. How can we boost butyrate-producing bacteria? Prebiotics, food ingredients that stimulate the growth and/or activity of bacteria in the digestive system may be the best option. Two such food components are inulin and fructooligosaccharides (FOS), which in vitro, stimulate growth of the butyrate producers F. prausnitzii, E.rectale, E. hallii and R. intestinalis 4-15 fold above basal levels (Scott et al. 2014). In vivo, consumption of 10g/day of inulin for 16 days in healthy, middle aged humans (BMI 25 kg*m-2, avg. age 38) significantly stimulated growth of F. prausnitzii (Ramirez-Farias et al. 2009). Therefore, consumption of foods rich in inulin and FOS may be a valid strategy for boosting levels of butyrate-producing bacteria in our intestines.
Foods containing inulin and fructoligosaccharides are shown in Table 1.
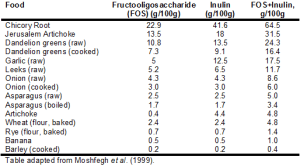
First, it’s important to mention that average intake for inulin and FOS is only ~2.5 grams/day (5g total) (Moshfegh et al. 1999). This is because relatively few foods contain high amounts of these prebiotic fibers. For example, whereas 100g of bananas (equivalent to 1 small banana) contains 1 gram total of combined inulin and FOS, in contrast, chicory root and Jerusalem artichoke contain 64.5 and 31.5 grams, respectively. In addition, although not shown in the table, most fruits contain limited amounts of FOS. Bananas contain more FOS/g than apple, blackberry, blueberry, cantaloupe, grapes, orange, peach, pear, raspberry, rhubarb, strawberry and watermelon (Dumitiriu 2005). This is an important finding because one would expect fruits to be rich in inulin and FOS, as both of these fibers contain long chain fructose polymers. Furthermore, based on values for asparagus, chicory, onion, loss of inulin and FOS upon boiling is ~30%, so eating these foods raw is not the only strategy for increasing dietary amounts of FOS and inulin.
Can increasing consumption of FOS and inulin improve health and lifespan? To date, dietary supplementation with inulin has been shown to improve cognitive performance (Messaoudi et al. 2005), and, to reduce cholesterol, triglycerides and body weight, and, improved survival in rats (Rozan et al. 2008). Although randomized controlled trials examining the effect of increasing butyrate-producing bacteria on health and mortality risk in older adults has yet to be performed, collectively, the evidence presented here suggests that if you’re interested in a low risk, potentially high reward approach towards improving health and lifespan, consuming more foods containing FOS and inulin may be a valid strategy!
If you’re interested, please have a look at my book!
References
Biagi E, Nylund L, Candela M, Ostan R, Bucci L, Pini E, Nikkïla J, Monti D, Satokari R, Franceschi C, Brigidi P, De Vos W. Through ageing, and beyond: gut microbiota and inflammatory status in seniors and centenarians. PLoS One. 2010 May 17;5(5):e10667.
Dumitiriu S. Polysaccharides: Structural Diversity and Functional Versatility. 2005. CRC Press. p854-855.
Hariri N, Gougeon R, Thibault L. A highly saturated fat-rich diet is more obesogenic than diets with lower saturated fat content. Nutr Res. 2010 Sep;30(9):632-43.
Harrison DE, Strong R, Allison DB, Ames BN, Astle CM, Atamna H, Fernandez E, Flurkey K, Javors MA, Nadon NL, Nelson JF, Pletcher S, Simpkins JW, Smith D, Wilkinson JE, Miller RA. Acarbose, 17-α-estradiol, and nordihydroguaiaretic acid extend mouse lifespan preferentially in males. Aging Cell. 2013 Oct 26.
Kaeberlein, M., McVey, M. and Guarente, L. (1999). The SIR2/3/4 complex and SIR2 alone promote longevity in Saccharomyces cerevisiae by two different mechanisms. Genes Dev. 13, 2570-2580.
Kang, H. L., Benzer, S. and Min, K. T. (2002). Life extension in Drosophila by feeding a drug. Genetics 99, 838-843. Kim, S., Benguria, A., Lai, C. and Jazwinski, S. M. (1999). Modulation of lifespan by histone deacetylase genes in Saccharomyces cerevisiae. Mol. Biol. Cell 10, 3125-3156.
Kurapati R, Passananti HB, Rose MR, Tower J. Increased hsp22 RNA levels in Drosophila lines genetically selected for increased longevity. J Gerontol A Biol Sci Med Sci. 2000 Nov;55(11):B552-9.
Li H, Gao Z, Zhang J, Ye X, Xu A, Ye J, Jia W. Sodium butyrate stimulates expression of fibroblast growth factor 21 in liver by inhibition of histone deacetylase 3. Diabetes. 2012 Apr;61(4):797-806.
McDonald P, Maizi BM, Arking R. Chemical regulation of mid- and late-life longevities in Drosophila. Exp Gerontol. 2013 Feb;48(2):240-9.
Messaoudi M, Rozan P, Nejdi A, Hidalgo S, Desor D. Behavioural and cognitive effects of oligofructose-enriched inulin in rats. Br J Nutr. 2005 Apr;93 Suppl 1:S27-30.
Moshfegh AJ, Friday JE, Goldman JP, Ahuja JK. Presence of inulin and oligofructose in the diets of Americans. J Nutr. 1999 Jul;129(7 Suppl):1407S-11S.
Morrow G, Samson M, Michaud S, Tanguay RM. Overexpression of the small mitochondrial Hsp22 extends Drosophila life span and increases resistance to oxidative stress. FASEB J. 2004 Mar;18(3):598-9.
Ramirez-Farias C, Slezak K, Fuller Z, Duncan A, Holtrop G & Louis P (2009) Effect of inulin on the human gut microbiota: stimulation of Bifidobacterium adolescentis and Faecalibacterium prausnitzii. Brit J Nutr 101: 533-542.
Rose MR. Can human aging be postponed? Sci Am. 1999 Dec;281(6):106-11.
Rozan P, Nejdi A, Hidalgo S, Bisson JF, Desor D, Messaoudi M. Effects of lifelong intervention with an oligofructose-enriched inulin in rats on general health and lifespan. Br J Nutr. 2008 Dec;100(6):1192-9.
Scott KP, Martin JC, Duncan SH, Flint HJ. Prebiotic stimulation of human colonic butyrate-producing bacteria and bifidobacteria, in vitro. FEMS Microbiol Ecol. 2014 Jan;87(1):30-40.
Tap J, Mondot S, Levenez F et al. (2009) Towards the human intestinal microbiota phylogenetic core. Environ Microbiol 11: 2574-2584.
Tissenbaum, H. A. and Guarente, L. (2001). Increased dosage of a sir2 gene extends lifespan in Caenorhabditis elegans. Nature 410, 227-230
Walker AW, Ince J, Duncan SH et al. (2011) Dominant and diet-responsive groups of bacteria within the human colonic microbiota. ISME J 5: 220-230.
Wolever TM, Chiasson JL. Acarbose raises serum butyrate in human subjects with impaired glucose tolerance. Br J Nutr. 2000 Jul;84(1):57-61.
Witt O, Deubzer HE, Milde T, Oehme I. HDAC family: What are the cancer relevant targets? Cancer Lett. 2009 May 8;277(1):8-21.
Zhang Y, Xie Y, Berglund ED, Coate KC, He TT, Katafuchi T, Xiao G, Potthoff MJ, Wei W, Wan Y, Yu RT, Evans RM, Kliewer SA, Mangelsdorf DJ. The starvation hormone, fibroblast growth factor-21, extends lifespan in mice. Elife. 2012;1:e00065.
Zhao Y, Sun H, Lu J, Li X, Chen X, Tao D, Huang W, Huang B. Lifespan extension and elevated hsp gene expression in Drosophila caused by histone deacetylase inhibitors. J Exp Biol. 2005 Feb;208(Pt 4):697-705.